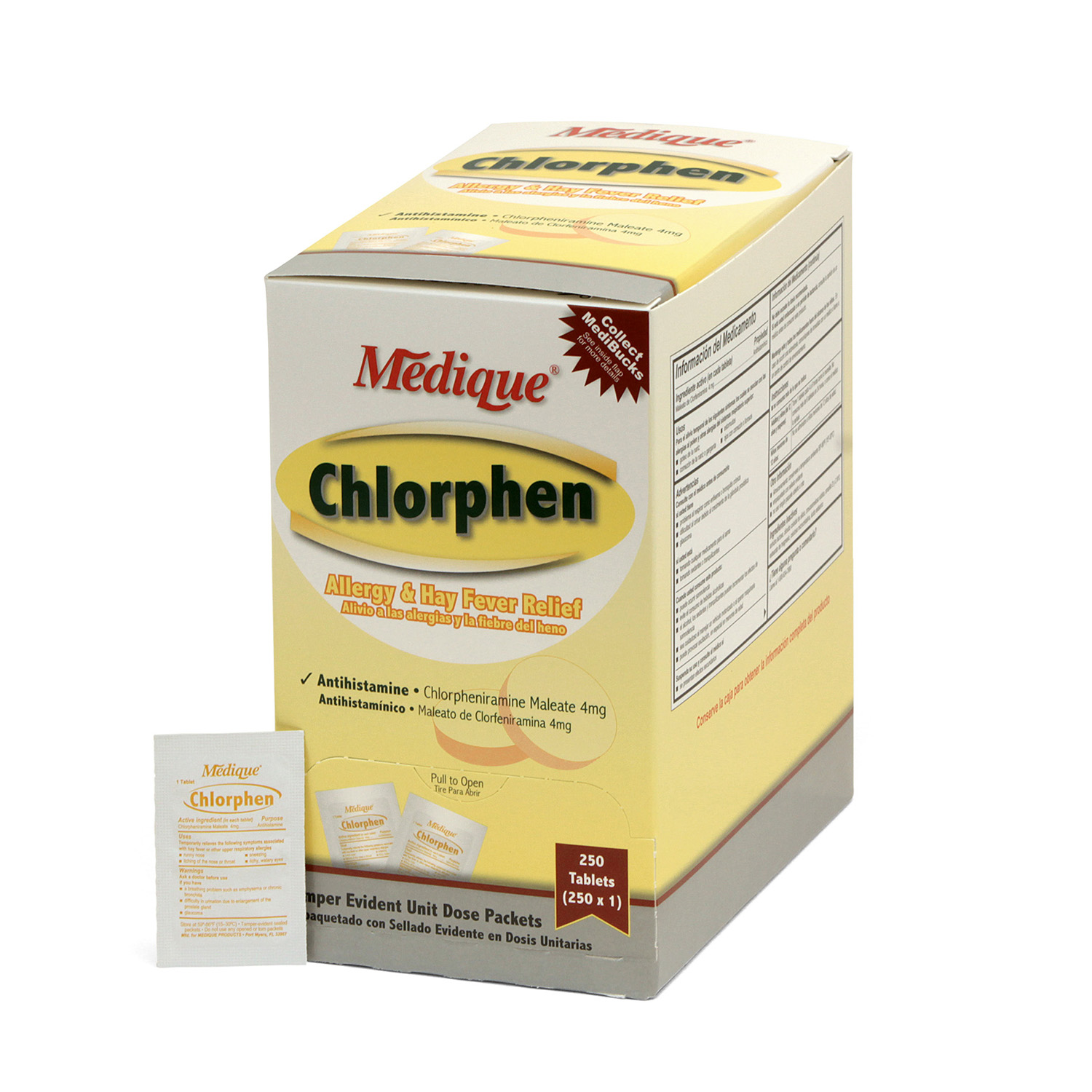
Chlorphen Tabs 250×1
Compare active ingredient to Chlor-Trimeton®
For relief from sneezing, itchy nose and throat, and watery eyes caused by hay fever and other symptoms of allergic reactions. May cause drowsiness.
Each tablet contains:
Chlorpheniramine Maleate 4mg
Drug Facts
Ingredients
Active ingredient (in each tablet): Chlorpheniramine Maleate 4 mg
Purpose:
Antihistamine
Inactive ingredients
corn starch, D&C yellow #10 aluminum lake, lactose anhydrous, magnesium stearate, microcrystalline cellulose
Uses
temporarily relieves these symptoms due to hay fever or other upper respiratory allergies:
■ runny nose
■ itchy, watery eyes
■ sneezing
■ itching of the nose or throat
Warnings
Do not use to make a child sleepy.
Ask a doctor before use if you have
■ glaucoma
■a breathing problem such as emphysema or chronic bronchitis
■ trouble urinating due to an enlarged prostate gland
Ask a doctor or pharmacist before use if you are taking sedatives or tranquilizers
When using this product
■ drowsiness may occur
■ avoid alcohol beverages
■ alcohol, sedatives and tranquilizers may increase drowsiness
■ use caution when driving a motor vehicle or operating machinery
■ excitability may occur, especially in children
If you are pregnant or breast-feeding, ask a health professional before use.
Keep out of reach of children. In case of overdose, get medical help or contact a Poison Control Center (1-800-222-1222) right away.
Directions
Adults and children: (12 years and older) 1 tablet every 4 to 6 hours. Do not take more than 6 tablets in 24 hours.
Children under 12 years: Do not give to children under 12 years of age.
Other Information
■ tamper-evident sealed packets
■ do not use any opened or torn packets
■ store at 77ºF (25ºC); excursions permitted between 59º-86ºF (15º-30ºC)
■ protect from excessive moisture
■ use by expiration date on packet
Questions? Please contact us at 1-800-634-7680
Medique Products takes great care to ensure that the product you take is completely safe. This website contains current product information as of 3/31/2023 and may differ from the information on the product packaging you may have.